Brain’s Molecular Double Agent: Protein “Eato” Unveiled as Unexpected Neuroprotector
Brain’s Molecular Double Agent: Protein “Eato” Unveiled as Unexpected Neuroprotector
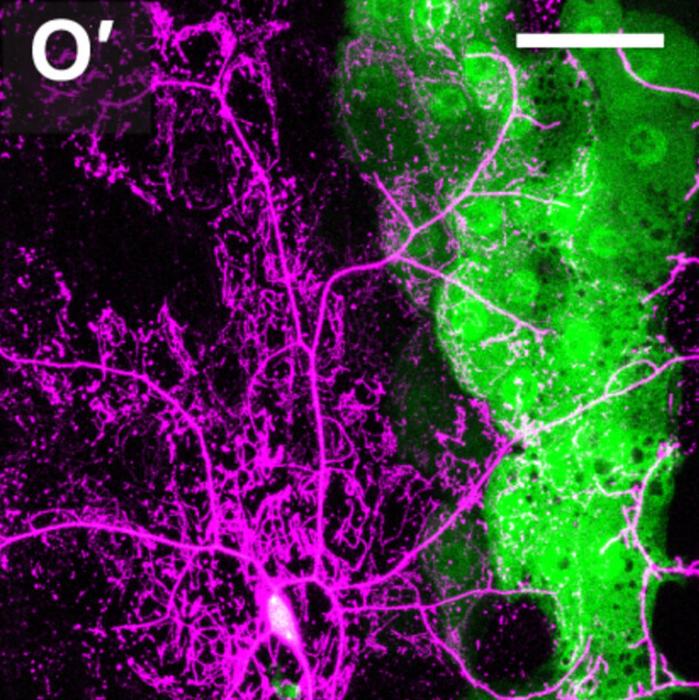
A groundbreaking discovery by researchers at Cornell University is reshaping our understanding of neurodegenerative diseases such as Alzheimer’s and Parkinson’s. Utilizing the fruit-fly model, Drosophila melanogaster, renowned for its biological similarities to human cellular processes, the team uncovered a novel function of the protein Eato, an ABCA transporter. Previously recognized for its role in lipid regulation within mammalian brain cells, Eato’s expanded role now reveals a dual, seemingly paradoxical function: protecting neurons while enhancing their clearance by phagocytic cells under specific conditions.
Eato belongs to the ABCA family of membrane transporters, proteins integral to maintaining lipid homeostasis by regulating the trafficking of phospholipids across cell membranes. Among these, human homologs such as ABCA1 and ABCA7 have established links to Alzheimer’s disease, underlining the clinical relevance of this protein family in neurodegeneration. The Cornell team’s insights emerged from observing the behavior of Eato-deficient neurons in fruit flies, revealing that neuron survival hinges on the delicate balance Eato maintains in the exposure of a critical "eat-me" signal—phosphatidylserine (PS).
Phosphatidylserine, a phospholipid normally localized to the inner leaflet of the plasma membrane, flips to the cell surface during neuronal stress or apoptosis, serving as an identification flag for phagocytes. These immune cells are responsible for the targeted clearance of dying or damaged neurons, a process that, if improperly regulated, can lead to excessive neuronal loss and exacerbate neurodegenerative pathology. The researchers demonstrated that Eato suppresses premature PS exposure on healthy neurons, thereby preventing their untimely engulfment by phagocytes.
Unexpectedly, the research revealed that removal of Eato from neurons resulted in increased PS surface exposure and subsequent neuron loss. Contrarily, elimination of Eato from phagocytic glial cells led to reduced neuronal degeneration, indicating the protein’s bifunctional influence. This duality suggests Eato not only modulates the suppression of PS exposure within neurons but also facilitates recognition of PS by phagocytes, orchestrating neuron-glia interactions fundamental to brain homeostasis. Such opposing roles compel a reevaluation of ABCA transporters’ functions in neurobiology.
The team utilized sophisticated live imaging and genetically engineered mutations in Drosophila to monitor PS exposure dynamics and phagocytic activity. They identified distinct phenotypic categories ranging from normal neural integrity to degeneration localized within specific brain regions, emphasizing the spatial precision of Eato’s action. Their findings illuminate formerly obscure mechanisms underpinning phagocytosis-driven neurodegeneration and open novel avenues for targeted therapeutic intervention.
Neurodegenerative diseases impose a monumental socioeconomic burden. Alzheimer’s disease alone accounts for approximately $305 billion in care costs annually in the United States, projected to exceed $1 trillion as global demographics shift toward aging populations. Parkinson’s disease similarly strains healthcare systems, with annual costs surpassing $50 billion due to direct medical expenditures and indirect impacts such as lost productivity and caregiver demands. Innovative treatments addressing underlying cellular mechanisms are urgently needed.
By elucidating how Eato governs PS exposure and neuronal fate, the Cornell study provides a credible molecular target for therapies aimed at halting or mitigating neurodegeneration. In principle, drugs designed to sustain appropriate PS localization or modulate phagocyte detection mechanisms might prevent the premature clearance of healthy neurons. This strategy contrasts with traditional approaches that primarily address symptomatic relief rather than disease causation.
Professor Chun Han, leading the research at Cornell’s Weill Institute for Cell and Molecular Biology, highlighted the novelty of these findings. “Eato’s dualistic regulation of PS exposure in both neurons and glia defies prior assumptions about ABCA transporter function,” he stated. This nuanced understanding challenges the simplistic model of PS as an unregulated apoptosis marker, underscoring an active maintenance system in healthy neuronal environments.
The implications extend beyond Alzheimer’s and Parkinson’s diseases, as aberrant phagocytosis is implicated in various neuroinflammatory and degenerative conditions. Understanding the molecular crosstalk between neurons and phagocytes mediated by lipid signals could redefine disease pathogenesis and guide precision medicine efforts.
Importantly, removing PS exposed on Eato-deficient neurons experimentally was found to prevent neuronal degeneration. This suggests that manipulation of lipid signaling pathways holds promise for neuroprotection, potentially allowing for preservation of cognitive and motor functions lost in these devastating disorders.
Moving forward, the study opens intriguing questions about how similar mechanisms operate in mammalian systems and how ABCA transporters coordinate complex lipid signaling across different brain cell types. The intricate balance between protecting neural tissue and enabling its clearance when damaged represents a crucial frontier in neuroscience research.
In summary, the Cornell team’s discovery of Eato’s opposing roles in neurons and phagocytes significantly advances our molecular understanding of neurodegeneration. By highlighting the importance of regulated phosphatidylserine exposure and subsequent phagocytic response, their work lays the foundation for innovative strategies aimed at protecting the brain’s vulnerable cells and slowing the progression of incurable diseases.
Other key contributors to this pioneering work include Bei Wang, Ankita Sarkar, Zixian Huang, Nicolas Vergara Ruiz, Ann T. Yeung, and Rachael Chen, all affiliated with Cornell University’s Weill Institute. Their collective efforts bring us closer to unlocking some of the brain’s most guarded secrets, holding hope for millions affected worldwide.
Subject of Research: Molecular mechanisms of neurodegeneration involving ABCA transporter Eato and phosphatidylserine exposure in neurons and phagocytes.
Article Title: Phagocytosis-driven neurodegeneration through opposing roles of an ABC transporter in neurons and phagocytes
News Publication Date: 12-Mar-2025
Web References:
- Science Advances article
- Han Lab at Cornell University
Image Credits: Han Lab
Keywords: Molecular biology, Neurodegeneration, Alzheimer’s disease, Parkinson’s disease, ABCA transporters, Phosphatidylserine, Phagocytosis, Neuron-glia interaction, Lipid signaling
Enzymatic Dual-Oxa Diels–Alder Builds Complex Acetal
Next PostMen Face Higher Risk and Lower Care-Seeking Rates for Three Common Diseases, Study Finds
New Benchmark Study Reveals Emerging Trends in Canine Behavior