Radioiodine Therapy Enhances Survival Outcomes in Differentiated Thyroid Cancer Patients
Radioiodine Therapy Enhances Survival Outcomes in Differentiated Thyroid Cancer Patients
Differentiated thyroid cancer represents one of the most commonly diagnosed endocrine malignancies worldwide. Over the decades, the use of radioactive iodine (RAI) therapy following surgical resection has been integral in management, particularly for high-risk patients. However, the effectiveness of RAI in improving long-term survival for patients with low- to intermediate-risk differentiated thyroid cancer has remained a topic of clinical uncertainty and debate. New research, published in the April 2025 issue of The Journal of Nuclear Medicine, has now provided compelling evidence through a large-scale retrospective analysis supporting the survival benefit of RAI therapy across different risk categories and histologic subtypes.
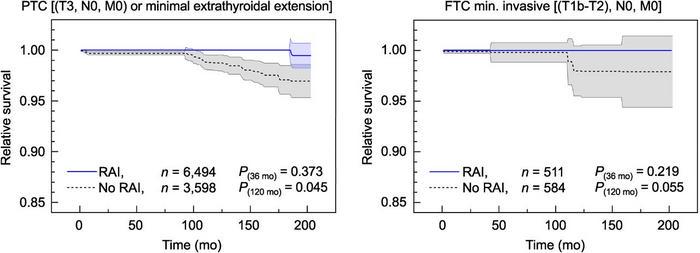
This comprehensive study leveraged real-world data from over 101,000 patients identified within the Surveillance, Epidemiology, and End Results Program (SEER) database, a robust nationwide cancer registry in the United States. The investigators stratified cohorts based on histological classification—classical papillary thyroid cancer (PTC), aggressive variants of PTC, follicular thyroid cancer (FTC), and minimally invasive FTC—and further categorized patients by their initial recurrence risk into very low, low, intermediate, and high risk. This stratification allowed for nuanced analysis of relative survival, which compares survival of cancer patients to that of matched individuals without cancer, offering a refined measure of treatment impact.
The findings reveal a significant survival advantage in patients who underwent RAI therapy across most subgroups, challenging prior clinical controversies. Particularly striking was the observed relative survival benefit of up to 30.9% in the high-risk differentiated thyroid cancer population, reaffirming the established therapeutic value of RAI in aggressive disease settings. Moreover, even in low- and intermediate-risk groups, where the role of RAI had been less clear, subtle but consistent trends favored RAI intervention. Classical PTC patients with larger tumors or lymph node metastases experienced a 1.3% to 2.0% increase in 10-year relative survival when treated with RAI. Notably, low-risk minimally invasive FTC patients showed a positive survival tendency, hinting that RAI’s protective effects may extend beyond traditionally indicated populations.
Mechanistically, radioactive iodine therapy exploits the physiological uptake of iodide by thyroid tissue, delivering targeted radiation to residual thyroid cancer cells or micrometastases post-thyroidectomy. This targeted cytotoxicity is critical in eradicating occult disease and preventing recurrence. While RAI use has been standardized for high-risk differentiated thyroid cancers, the heterogeneity in tumor biology and disease course in lower-risk groups has fueled divergent clinical guidelines and practices. This study’s extensive evaluation of histologic subtypes and risk categories helps bridge these gaps, providing data-driven insights to inform precision medicine approaches.
It is also noteworthy that the research indicates no detrimental survival effect associated with RAI treatment in any subgroup analyzed. This finding addresses important safety considerations and risk-benefit assessments clinicians must undertake when recommending adjuvant therapies. Furthermore, the improved survival trends become more pronounced approximately eight years after treatment, emphasizing the necessity for long-term follow-up in thyroid cancer survivorship studies.
The implications of these results extend into clinical decision-making and guideline development. As Dr. Henning Weis, lead investigator and nuclear medicine physician at University Hospital Cologne, emphasized, real-world evidence derived from large datasets such as SEER can be pivotal in resolving controversies where randomized controlled trials might be impractical. These insights are particularly valuable given the slow-growing nature of differentiated thyroid cancer and the challenges in accruing long-term survival data.
Additionally, co-author Professor Matthias Schmidt highlighted the substantial investment of nearly a decade in developing comprehensive thyroid cancer treatment guidelines. The current analysis represents a cornerstone, providing empirical substantiation on the survival impact of RAI therapy across diverse patient populations. These findings empower nuclear medicine and endocrine specialists to tailor treatment plans more confidently, balancing potential benefits with clinical nuances inherent to individual cases.
Beyond survival benefits, RAI therapy’s role in reducing recurrence rates has been well established in high-risk differentiated thyroid cancers. This study’s confirmation of survival advantages underscores the dual impact of RAI on disease control and mortality outcomes. Importantly, the research suggests a paradigm shift in considering adjuvant RAI therapy even in patients with low- or intermediate-risk profiles, provided a thorough assessment of tumor characteristics and patient-specific factors.
While the study’s retrospective nature and reliance on registry data introduce certain limitations inherent to observational analyses, its scale, methodological rigor, and comprehensive risk stratification enhance the robustness of the conclusions. Ongoing research integrating molecular and genetic tumor profiling alongside clinical parameters will likely further refine individualized therapeutic strategies in differentiated thyroid cancer.
In conclusion, this landmark investigation delineates a clear survival advantage conferred by radioactive iodine therapy following surgical intervention in differentiated thyroid cancer patients across various histologic subtypes and risk groups. It challenges existing paradigms, especially concerning low- to intermediate-risk cases, and provides a valuable evidence base to guide clinical practice in nuclear medicine and endocrinology. The balance between maximizing therapeutic benefit while minimizing overtreatment remains critical, but these findings mark a significant advance in understanding RAI therapy’s role within precision oncology for thyroid cancer.
Subject of Research: Impact of radioactive iodine treatment on long-term relative survival in differentiated thyroid cancer patients stratified by histologic subtypes and recurrence risk categories.
Article Title: Open Access Impact of Radioactive Iodine Treatment on Long-Term Relative Survival in Patients with Papillary and Follicular Thyroid Cancer: A SEER-Based Study Covering Histologic Subtypes and Recurrence Risk Categories
News Publication Date: April 1, 2025
Web References:
- DOI link to the original article
- The Journal of Nuclear Medicine (JNM)
References:
Weis H, Weindler J, Schmidt K, Drzezga A, Schmidt M, Hellmich M. Impact of Radioactive Iodine Treatment on Long-Term Relative Survival in Patients with Papillary and Follicular Thyroid Cancer: A SEER-Based Study Covering Histologic Subtypes and Recurrence Risk Categories. J Nucl Med. 2025 Apr; (Epub ahead of print).
Image Credits: Images created by Henning Weis, PhD, MD, and Prof. Matthias Schmidt, MD, FEBNM, Department of Nuclear Medicine, University Hospital of Cologne.
Keywords: Thyroid cancer, Radioiodine therapy, Differentiated thyroid cancer, Papillary thyroid cancer, Follicular thyroid cancer, Relative survival, SEER database, Nuclear medicine, Cancer treatment, Precision medicine, Endocrine oncology, Radioactive iodine, Long-term survival
Researchers Unveil Innovative Approach to Boost Water Oxidation Catalysis
Next PostNew Initiative Funded by the German Research Foundation Set to Harness Unconventional Magnetism for Innovative IT Applications
AI Matches Dermatologists in Accuracy of Skin Cancer Assessments