Scientists Uncover Origins of Common Lung Cancer Linked to Smoking
Scientists Uncover Origins of Common Lung Cancer Linked to Smoking
A newly published study has unveiled critical insights into the cellular origins and early progression of lung squamous cell carcinoma (LUSC), the second most prevalent form of lung cancer worldwide. Conducted by an interdisciplinary team from University College London (UCL), the Wellcome Sanger Institute, and the University of Cambridge, the research identifies a specific basal stem cell population in the trachea that becomes aberrantly dominant and drives carcinogenesis. This breakthrough paves the way for potential strategies focusing on earlier detection and preventive interventions in lung cancer, which remains the leading cause of cancer mortality globally.
Lung squamous cell carcinoma typically develops after prolonged exposure to inhaled carcinogens, most notably tobacco smoke, which induces cumulative genetic damage in the airway epithelium. However, understanding the specific cellular dynamics and early molecular events behind the transition from normal epithelium to precancerous lesions has remained elusive. This study illuminates a clonal expansion phenomenon originating from basal cells that express the Krt5 gene, responsible for producing a structural protein vital to epithelial integrity.
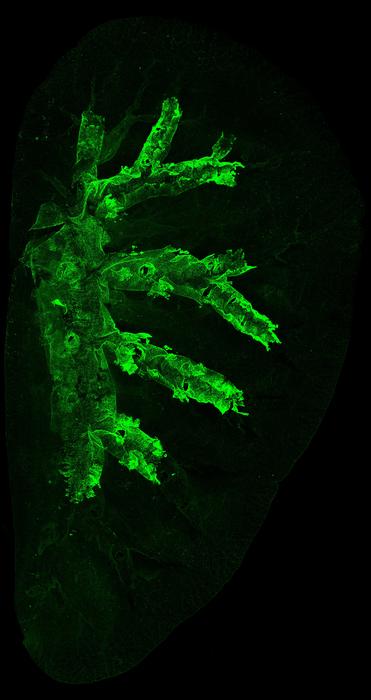
The research team leveraged sophisticated lineage tracing methods in genetically engineered mouse models to fluorescently label Krt5-positive basal cells in the trachea. By tracking these cells and their progeny over time, they observed that in mice exposed to carcinogens, a small subset of these basal cells multiply excessively and invade deeper lung tissue, displacing normal cells. In stark contrast, control mice without carcinogen exposure demonstrated stable basal cell populations confined to their native locations within the trachea.
Single-cell RNA sequencing further revealed that as these basal cells expand pathologically, they induce a shift in the cellular composition of the airway epithelium. Notably, a distinct population of transitional cells expressing Krt13 emerged, while some differentiated luminal cell subtypes became depleted. This alteration in cell type abundance signifies a disrupted equilibrium critical for maintaining normal airway function, suggesting that the pathological expansion of basal cells disturbs airway homeostasis to create a microenvironment conducive to cancer initiation.
Corroborating the murine data, DNA sequencing analyses of human lung and tracheal tissues from current and former smokers exposed to carcinogens demonstrated a clonally related origin of multiple precancerous lesions within and across both lungs. This finding underscores a paradigm where independent tumor foci arise from a singular damaged basal cell ancestor that disseminates and colonizes lung tissues, reinforcing the concept of basal cells as the crucial ‘cell of origin’ in LUSC pathogenesis.
The implications of these findings extend beyond mere cellular identification. By deciphering the competitive clonal dynamics and the cellular hierarchies that govern airway remodeling under carcinogen pressure, the research amplifies our understanding of early carcinogenesis. Tumorigenic basal cells exploit environmental niches within the lung epithelia where spatial and molecular conditions favor their genetic diversification and unchecked proliferation, eventually leading to tumor formation.
From a clinical perspective, identifying these aberrantly expanding basal cell clones and their associated cellular signatures offers an unprecedented window for earlier intervention. Current lung cancer diagnostics often detect the disease at advanced stages when treatment options are limited and prognosis poor. The discovery of precursor cellular states and their molecular markers holds promise for developing minimally invasive screening assays capable of identifying at-risk individuals before overt cancers develop.
Moreover, the study suggests novel preventive therapeutic avenues targeting the regulation of basal cell clonal expansion and the restoration of epithelial cellular balance. By modulating signaling pathways that underpin basal cell dominance or promoting the survival and function of protective luminal cells, it may become feasible to halt carcinogenesis at its root. Such strategies could dramatically alter the long-term outcomes of patients predisposed to LUSC, particularly habitual smokers.
Experts involved highlight that understanding the interplay between genetics and the lung microenvironment is crucial. The permissive niches enabling basal cell proliferation are shaped not only by intrinsic genetic mutations but also by extrinsic spatial factors within lung tissue architecture. Targeting this multifaceted crosstalk provides an integral framework for the development of next-generation therapies designed to prevent tumor initiation as well as progression.
This study exemplifies the power of combining experimental mouse models, single-cell transcriptomics, and high-resolution genetic analyses of human tissues to unravel complex disease mechanisms. It marks a significant advance in cancer biology by precisely delineating the first cellular events that usher in LUSC. As the world grapples with the burden of lung cancer, such fundamental insights forge a hopeful path toward earlier diagnosis, improved patient survival, and eventually, prevention.
In conclusion, the identification of Krt5-expressing basal cells in the trachea as the cell of origin for squamous cell lung cancer rewrites crucial chapters in our understanding of lung carcinogenesis. The detailed characterization of how these cells become clonally dominant and reshape airway epithelia opens new horizons in both research and clinical management. Continued investigations building on these foundations will be instrumental in translating this knowledge into tangible health benefits, potentially transforming outcomes in one of the deadliest cancers.
Subject of Research: Animals
Article Title: Aberrant basal cell clonal dynamics shape early lung carcinogenesis
News Publication Date: 1-May-2025
Web References: http://dx.doi.org/10.1126/science.ads9145
Image Credits: UCL
Keywords: Cancer, Squamous cell lung cancer
Registration and Scientific Program Now Open for Atomic, Molecular, and Optical Physics Conference
Next PostHKUST and PolyU Researchers Develop Innovative Laminate Structure to Enhance Perovskite Solar Cell Performance
AI Matches Dermatologists in Accuracy of Skin Cancer Assessments